Polytetrafluoroethylene (polytetrafluoroethylene) is probably the most widely used fluoropolymer because it has several characteristics that make it an ideal material for a wide range of applications. It is more flexible than other similar pipes and can resist almost all industrial chemicals
The temperature range is approximately -330°F to 500°F, providing the widest temperature range among fluoropolymers. In addition, it has excellent electrical properties and low magnetic permeability. Ptfe tubing is the most widely used laboratory tubing and applications where chemical resistance and purity are essential. PTFE has a very low coefficient of friction and is one of the most "slip" substances known
Features:
100% pure PTFE resin
Compared with FEP, PFA, HP PFA, UHP PFA, ETFE, ECTFE, most flexible fluoropolymer pipes
Chemically inert, resistant to almost all industrial chemicals and solvents
Wide temperature range
Low penetration
Smooth non-stick surface finish
Lowest friction coefficient
Excellent electrical performance
Non-flammable
Non-toxic
Applications:
laboratory
Chemical process
Analysis and process equipment
Emission monitoring
Low temperature
high temperature
Electricity
ozone
The structure of PTFE molecules
Polytetrafluoroethylene (PTFE) is made by the polymerization of many tetrafluoroethylene molecules
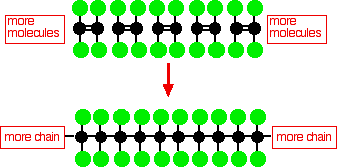
This simple PTFE diagram does not show the three-dimensional structure of the molecule. In the simpler molecular poly(ethylene), the carbon backbone of the molecule is connected only by hydrogen atoms, and this chain is very flexible-it is definitely not a linear molecule
However, in polytetrafluoroethylene, the fluorine atom in a CF2 group is large enough to interfere with the fluorine atom on the adjacent group. You have to remember that every fluorine atom has 3 pairs of lone electrons sticking out
The effect of this is to suppress the rotation of the carbon-carbon single bond. The fluorine atoms tend to be arranged so as to be as far away as possible from the adjacent fluorine atoms. Rotation tends to involve lone-pair collisions between fluorine atoms on adjacent carbon atoms-which makes the rotation energetically unfavorable
The repulsive force locks the molecule into a rod shape, and the fluorine atoms are arranged in a very gentle spiral—the fluorine atoms are arranged in a spiral around the carbon backbone. These lead strips will be squeezed together like long, thin pencils in a box
This close contact arrangement has an important influence on the intermolecular forces, as you will see
Intermolecular forces and the melting point of PTFE
The melting point of polytetrafluoroethylene is quoted as 327°C. This is quite high for this polymer, so there must be considerable van der Waals forces between the molecules
Why do people claim the van der Waals forces in PTFE are weak?
The van der Waals dispersion force is caused by the temporary fluctuating dipoles generated when the electrons in the molecule move around. Because the PTFE molecule is large, you would expect a large dispersion force because there are a lot of electrons that can move
The general situation is that the larger the molecule, the greater the dispersion power
However, PTFE has a problem. Fluorine is very electronegative. It tends to bind the electrons in the carbon-fluorine bond tightly together, so tightly that the electrons cannot move as you think. We describe the carbon-fluorine bond as not having strong polarization
Van der Waals forces also include dipole-dipole interactions. But in polytetrafluoroethylene (PTFE), each molecule is surrounded by a layer of slightly negatively charged fluorine atoms. In this case, the only possible interaction between molecules is mutual repulsion!
So the dispersion force is weaker than you think, and the dipole-dipole interaction will cause repulsion. No wonder people say that the van der Waals force in PTFE is very weak. You won't actually get the repulsive force, because the influence of the dispersion force is greater than that of the dipole-dipole interaction, but the net effect is that the van der Waals force will tend to weaken
But PTFE has a very high melting point, so the force that holds the molecules together must be very strong
How can PTFE have a high melting point?
PTFE is very crystalline, in this sense there is a large area, the molecules are in a very regular arrangement. Remember, PTFE molecules can be thought of as elongated rods. These poles will be closely clustered together
This means that although the ptfe molecule cannot produce really large temporary dipoles, the dipoles can be used very efficiently
So are the van der Waals forces in PTFE weak or strong?
I think you can be both right! If the polytetrafluoroethylene (PTFE) chains are arranged in such a way that there is not too close contact between the chains, the force between them will be very weak and the melting point will be very low
But in the real world, molecules are in close contact. Van der Waals forces may not be as powerful as they may be, but the structure of PTFE means they feel the greatest effect, producing overall strong intermolecular bonds and high melting points
This is in contrast to other forces, such as the dipole-dipole interaction force, which is only reduced by 23 times, or twice the distance is reduced by 8 times
Therefore, the tight packing of rod-shaped molecules in PTFE maximizes the effectiveness of dispersion
The non-stick properties
This is why water and oil do not stick to the surface of PTFE, and why you can fry eggs in a PTFE-coated pan without sticking to the pan
You need to consider what forces might fix other molecules on the surface of PTFE. It may include some kind of chemical bond, van der Waals force or hydrogen bond
Chemical bonding
The carbon-fluorine bond is very strong, and it is impossible for any other molecules to reach the carbon chain to cause any substitution reaction to take place. It is impossible for a chemical bond to occur
van der Waals forces
We have seen that the van der Waals force in PTFE is not very strong, and it will only make PTFE have a high melting point, because the molecules are so close that they have very effective contact.
But it is different for other molecules close to the surface of PTFE. Relatively small molecules (such as water molecules or oil molecules) will only have a small amount of contact with the surface, and only a small amount of van der Waals attraction will be generated.
A large molecule (such as a protein) will not be rod-shaped, so there is not enough effective contact between it and the surface to overcome the low polarization tendency of PTFE.
Either way, the van der Waals force between the surface of the PTFE and the surrounding things is small and ineffective
Hydrogen bonds
The PTFE molecules on the surface are completely wrapped by fluorine atoms. These fluorine atoms are very electronegative, so they all carry a certain degree of negative charge. Each fluorine also has 3 pairs of protruding lone electrons
These are the conditions required for the formation of hydrogen bonds, such as the lone pair on fluorine and the hydrogen atom in water. But this obviously will not happen, otherwise there will be a strong attraction between the PTFE molecules and the water molecules, and the water will stick to the PTFE
Summary
There is no effective way for other molecules to successfully attach to the surface of PTFE, so it has a non-stick surface
The low friction
The coefficient of friction of PTFE is very low. This means that if you have a surface coated with ptfe, other things will easily slip on it.
Below is a quick summary of what is happening. This comes from a 1992 paper entitled "Friction and Wear of Polytetrafluoroethylene".
At the beginning of the sliding, the PTFE surface breaks and the mass is transferred to wherever it is sliding. This means that the PTFE surface will wear.
As the sliding continued, the blocks unfolded into thin films.
At the same time, the surface of the PTFE is pulled out to form an organized layer.
Both surfaces in contact now have well-organized PTFE molecules that can slide on each other
The above is the introduction of polytetrafluoroethylene, polytetrafluoroethylene can be made into a variety of products, we are specialized in making ptfe tube、ptfe hose manufacturers, welcome to communicate with us
Searches related to ptfe hose:
Post time: May-05-2021





